PERIPHERAL JOINTS#
EFFICACY
OUTCOMES
ACROSS KEY PsA DOMAINS
INDICATION
RINVOQ is indicated for the treatment of adults with active psoriatic arthritis (PsA) who have had an inadequate response or intolerance to one or more tumor necrosis factor (TNF) blockers.
Limitations of Use: RINVOQ is not recommended for use in combination with other Janus kinase (JAK) inhibitors, biologic disease-modifying antirheumatic drugs (bDMARDs), or with potent immunosuppressants such as azathioprine and cyclosporine.
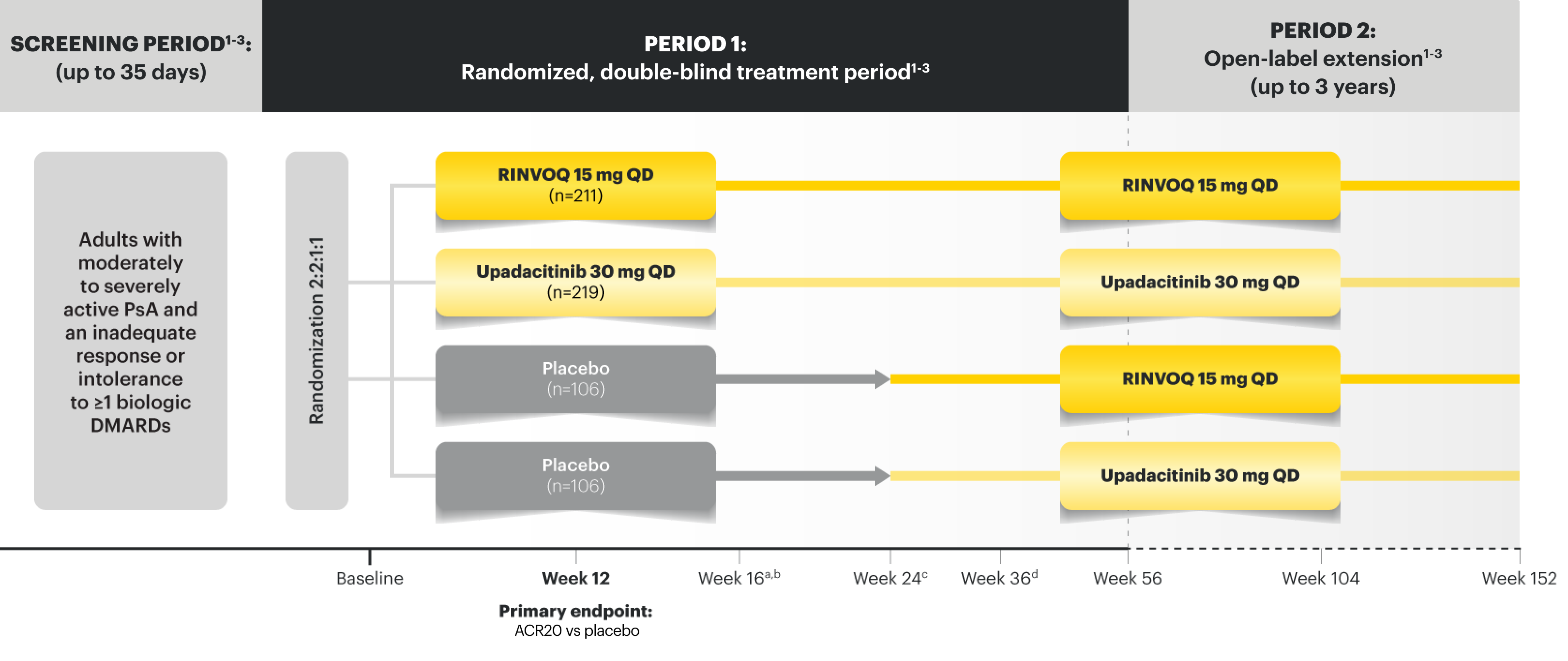
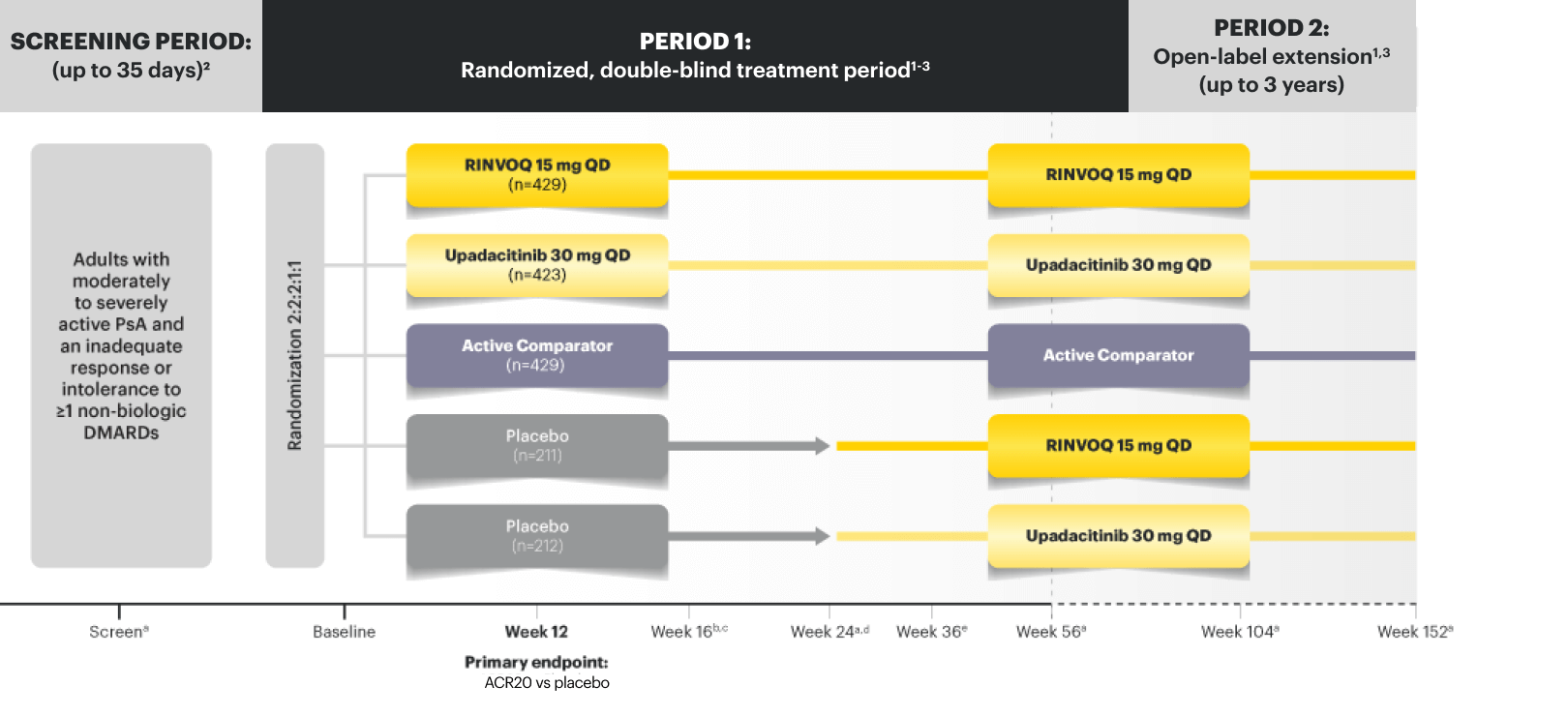
NRI Data From SELECT-PsA 2 (bDMARD-IR)1,2
RINVOQ 15 mg (n=211), placebo (n=212)
ACR20 | PRIMARY ENDPOINT
57%* RINVOQ vs 24% placebo at Week 12
*P<0.0012
SELECT-PsA 2 Study Design Intro: 24-week, double‑blind, placebo‑controlled study of 642 adult patients with moderately to severely active PsA who had an inadequate response or intolerance to at least 1 biologic DMARD. Patients were randomized to receive upadacitinib or placebo. The primary endpoint was proportion of patients achieving ACR20 response at Week 12 vs placebo.1
Please see Important Safety Information, including BOXED WARNING on Serious Infections, Mortality, Malignancies, Major Adverse Cardiovascular Events, and Thrombosis, below.
NRI Data From SELECT-PsA 2 (bDMARD-IR)2-4
ACR50
32%* RINVOQ (n=211) vs
5% placebo (n=212) at Week 12
Dactylitis Resolution (LDI=0)‡
58%* RINVOQ (n=55) vs
28% placebo (n=64) at Week 24
Enthesitis Resolution (LEI=0)§
43%* RINVOQ (n=133) vs
15% placebo (n=144) at Week 24
PASI 75 | RANKED SECONDARY ENDPOINT||
52%† RINVOQ (n=130) vs
16% placebo (n=131) at Week 16
BASDAI50¶
32%* RINVOQ (n=76) vs
4% placebo (n=75) at Week 24
DATA LIMITATIONS: Data labeled as a primary or ranked secondary endpoint were multiplicity-controlled for comparisons. All other comparisons were not adjusted for multiplicity; statistical significance has not been established.
*P<0.001; analyses were not controlled for multiplicity; P-value obtained through nominal statistical testing.2
†P<0.0012
SELECT-PsA 2: bDMARD-IR patients
ALL DATA ARE AS OBSERVED


60%
of PsA patients achieved ACR50
at Week 152 (OLE) (n=139)

DACTYLITIS
94%
of PsA patients achieved complete resolution
of dactylitis (LDI=0‡) at Week 152 (OLE) (n=36)

ENTHESITIS
69%
of PsA patients achieved complete resolution
of enthesitis (LEI=0§) at Week 152 (OLE) (n=88)

SKIN
66%
of PsA patients achieved skin clearance**
(PASI 75||) at Week 152 (OLE) (n=89)

AXIAL
63%
of PsA patients achieved BASDAI50¶ response
rates at Week 152 (OLE) (n=46)
DATA LIMITATIONS: BASDAI50 was a prespecified nonranked
endpoint and was not adjusted for multiplicity. Therefore, no statistical
or clinical conclusions can be made.
**RINVOQ is not indicated for the treatment of plaque psoriasis.
‡For patients with baseline presence of dactylitis (LDI>0).
§For patients with baseline presence of enthesitis (LEI>0).
||PASI assessed in patients with psoriatic skin involvement of ≥3% BSA at baseline.
¶For patients with presence of psoriatic spondylitis as assessed by the investigator.
#PsA patients in the SELECT-PsA 2 trial presented with peripheral arthritis, which manifested as tender or swollen peripheral joints.
The 2021 GRAPPA (Group for Research and Assessment of Psoriasis and Psoriatic Arthritis) guidelines recommend a domain-based approach for assessing psoriatic arthritis (PsA).8
As Observed (AO) analysis: Patients with missing data at a specific time are not included, which may enrich the population and increase the response rates.
OLE LIMITATIONS: There is potential for enrichment of OLE data; unblinding patients may cause bias related to the overall treatment effect.
‡For patients with baseline presence of dactylitis (LDI>0).
§For patients with baseline presence of enthesitis (LEI>0).
||PASI assessed in patients with psoriatic skin involvement of ≥3% BSA at baseline.
¶For patients with presence of psoriatic spondylitis as assessed by the investigator.
#PsA patients in the SELECT-PsA 2 trial presented with peripheral arthritis, which manifested as tender or swollen peripheral joints.

OF RINVOQ PATIENTS ACHIEVED MDA vs 3% (n=6/212) placebo at
Week 24 (NRI) (ranked secondary endpoint, P<0.001)²
Nearly Half (44%) of RINVOQ Patients Achieved MDA at ~3 Years9
SELECT-PsA 2: bDMARD-IR patients9
ALL DATA ARE AS OBSERVED


OF RINVOQ PATIENTS ACHIEVED MDA vs 3% (n=6/212) placebo at
Week 24 (NRI) (ranked secondary endpoint, P<0.001)²
As Observed (AO) analysis: Patients with missing data at a specific time are not included, which may enrich the population and increase the response rates.
OLE LIMITATIONS: There is potential for enrichment of OLE data; unblinding patients may cause bias related to the overall treatment effect.
Safety Considerations
Serious Infections: RINVOQ-treated patients are at increased risk of serious bacterial (including tuberculosis [TB]), fungal, viral, and opportunistic infections leading to hospitalization or death. Most patients who developed these infections were taking concomitant immunosuppressants, such as methotrexate or corticosteroids.
Mortality: A higher rate of all-cause mortality, including sudden cardiovascular (CV) death, was observed with a Janus kinase inhibitor (JAKi) in a study comparing another JAKi with tumor necrosis factor (TNF) blockers in rheumatoid arthritis (RA) patients ≥50 years with ≥1 CV risk factor.
Malignancies: Malignancies have occurred in RINVOQ-treated patients. A higher rate of lymphomas and lung cancer (in current or past smokers) was observed with another JAKi when compared with TNF blockers in RA patients.
Major Adverse Cardiovascular Events: A higher rate of CV death, myocardial infarction, and stroke was observed with a JAKi in a study comparing another JAKi with TNF blockers in RA patients ≥50 years with ≥1 CV risk factor. History of smoking increases risk.
Thromboses: Deep venous thrombosis, pulmonary embolism, and arterial thrombosis have occurred in patients treated for inflammatory conditions with JAK inhibitors, including RINVOQ. A higher rate of thrombosis was observed with another JAKi when compared with TNF blockers in RA patients.
Hypersensitivity: RINVOQ is contraindicated in patients with hypersensitivity to RINVOQ or its excipients.
Other Serious Adverse Reactions: Hypersensitivity Reactions, Gastrointestinal Perforations, Laboratory Abnormalities, and Embryo-Fetal Toxicity.
Durable Joint Efficacy in PsA2,4,9
ACR20/50/70: Durable Joint Response Rates Out to ~3 Years
NRI Data From SELECT-PsA 2 (bDMARD-IR)2
RINVOQ 15 mg (n=211), placebo (n=212)
ACR20 | PRIMARY ENDPOINT
57%* RINVOQ vs 24% placebo
at Week 12
ACR50
32%† RINVOQ vs 5% placebo
at Week 12
ACR70
9%† RINVOQ vs 1% placebo
at Week 12
DATA LIMITATIONS: Data labeled as a primary or ranked secondary endpoint were multiplicity-controlled for comparisons. All other comparisons were not adjusted for multiplicity; statistical significance has not been established.
*P<0.0012
†P<0.001; analyses were not controlled for multiplicity; P-value obtained through nominal statistical testing.2
NRI Data From SELECT-PsA 24
RINVOQ (n=211), Placebo (n=212)
RAPID ACR20 RESPONSES
33% vs 11% placebo* at Week 2
57% vs 24% placebo at Week 12 PRIMARY ENDPOINT
ACR50
32% vs 5% placebo* at Week 12
ACR70
9% vs 1% placebo* at Week 12
*P<0.001; analyses were not controlled for multiplicity; P-value obtained through nominal statistical testing4
†P<0.0014
RAPID RESPONSE

FOR ACR20 (NRI)
SELECT-PsA 2: bDMARD-IR patients9
ALL DATA ARE AS OBSERVED

RAPID RESPONSE

FOR ACR20 (NRI)
As Observed (AO) analysis: Patients with missing data at a specific time are not included, which may enrich the population and increase the response rates.
OLE LIMITATIONS: There is potential for enrichment of OLE data; unblinding patients may cause bias related to the overall treatment effect.
Complete Resolution of Enthesitis and Dactylitis Data4
NRI Data From SELECT-PsA 2 (bDMARD-IR)4
RINVOQ 15 mg (n=133), placebo (n=144)
Enthesitis Resolution (LEI=0)‡
43%|| RINVOQ vs 15% placebo at Week 24
NRI Data From SELECT-PsA 2 (bDMARD-IR)4
RINVOQ 15 mg (n=55), placebo (n=64)
Dactylitis Resolution (LDI=0)§
58%|| RINVOQ vs 28% placebo at Week 24
DATA LIMITATIONS: Data labeled as a primary or ranked secondary endpoint were multiplicity-controlled for comparisons. All other comparisons were not adjusted for multiplicity; statistical significance has not been established.
||P≤0.05; analyses were not controlled for multiplicity; P-value obtained through nominal statistical testing.4
SELECT-PsA 2: bDMARD-IR patients9
ALL DATA ARE AS OBSERVED

‡For subjects with baseline presence of enthesitis (LEI>0).
§For subjects with baseline presence of dactylitis (LDI>0).
As Observed (AO) analysis: Patients with missing data at a specific time are not included, which may enrich the population and increase the response rates.
OLE LIMITATIONS: There is potential for enrichment of OLE data; unblinding patients may cause bias related to the overall treatment effect.
Durable HAQ-DI Responses for RINVOQ Up to ~3 Years6
IMPROVEMENT IN
PHYSICAL FUNCTION
SELECT-PsA 2 MMRM DATA4
LS MEAN ∆HAQ-DI
RANKED SECONDARY ENDPOINT
Week 12: -0.30 vs -0.10 placebo¶
RINVOQ (n=199), placebo (n=180)
¶P<0.001
SELECT-PsA 2: bDMARD-IR patients6
ALL DATA ARE AS OBSERVED

IMPROVEMENT IN
PHYSICAL FUNCTION
SELECT-PsA 2 MMRM DATA4
LS MEAN ∆HAQ-DI
RANKED SECONDARY ENDPOINT
Week 12: -0.30 vs -0.10 placebo¶
RINVOQ (n=199), placebo (n=180)
¶P<0.001
As Observed (AO) analysis: Patients with missing data at a specific time are not included, which may enrich the population and increase the response rates.
OLE LIMITATIONS: There is potential for enrichment of OLE data; unblinding patients may cause bias related to the overall treatment effect.
Please see Important Safety Information, including BOXED WARNING on Serious Infections, Mortality, Malignancies, Major Adverse Cardiovascular Events, and Thrombosis, below.
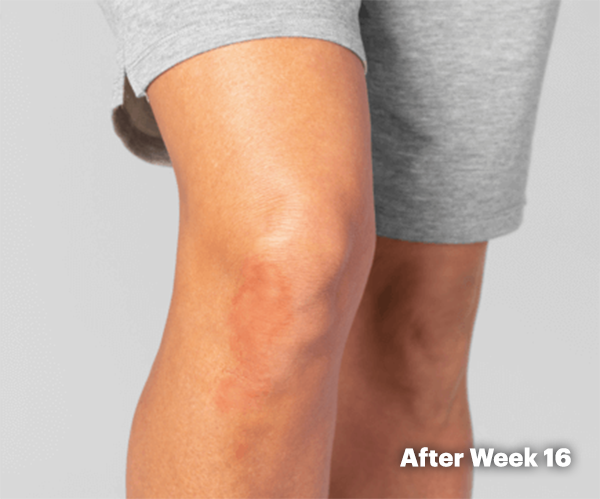
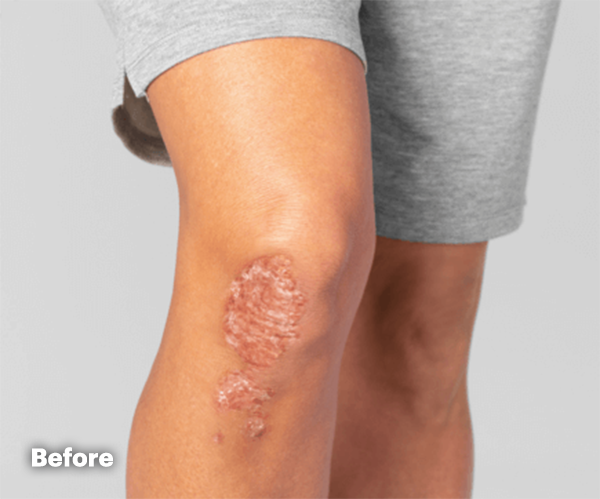
PsA Skin Clearance Before and After
In SELECT-PsA 2, 52% of RINVOQ patients achieved PASI 75 vs 16% placebo at Week 16
(ranked secondary endpoint, NRI, P<0.001)2
Drag the slider below to see results.
Illustration of PASI assessed in patient with
psoriatic skin involvement ≥3% BSA at baseline
Illustration of PASI assessed in patient with
psoriatic skin involvement ≥3% BSA at baseline
RINVOQ has not been studied in and is not indicated for the treatment of plaque psoriasis.1
Skin Clearance Data Through ~3 Years7,9*§
NRI Data From SELECT-PsA 2 (bDMARD-IR)2
RINVOQ 15 mg (n=130), placebo (n=131)
PASI 75* | RANKED SECONDARY ENDPOINT
52%† RINVOQ vs 16% placebo
at Week 16
PASI 90*
35%‡ RINVOQ vs 8% placebo
at Week 16
PASI 100*
25%‡ RINVOQ vs 6% placebo
at Week 16
DATA LIMITATIONS: Data labeled as a primary or ranked secondary endpoint were multiplicity-controlled for comparisons. All other comparisons were not adjusted for multiplicity; statistical significance has not been established.
RINVOQ is not indicated for the treatment of plaque psoriasis.1
*PASI assessed in patients with psoriatic skin involvement of ≥3% BSA at baseline.2
†P<0.0012
‡P<0.001; analyses were not controlled for multiplicity; P-value obtained through nominal statistical testing.2
NRI Data From SELECT-PsA 24
RINVOQ (n=211), Placebo (n=212)
RAPID ACR20 RESPONSES
33% vs 11% placebo* at Week 2
57% vs 24% placebo at Week 12 PRIMARY ENDPOINT
ACR50
32% vs 5% placebo* at Week 12
ACR70
9% vs 1% placebo* at Week 12
*P<0.0012
†PASI assessed in patients with psoriatic skin involvement of ≥3% BSA at baseline2,4
SELECT-PsA 2: bDMARD-IR patients9
ALL DATA ARE AS OBSERVED

*PASI assessed in patients with psoriatic skin involvement of ≥3% BSA at baseline.2
§After Week 16, the use of concomitant treatments for psoriasis was permitted.2
RINVOQ is not indicated for the treatment of plaque psoriasis.1
As Observed (AO) analysis: Patients with missing data at a specific time are not included, which may enrich the population and increase the response rates.
OLE LIMITATIONS: There is potential for enrichment of OLE data; unblinding patients may cause bias related to the overall treatment effect.

Interested in the safety data for RINVOQ?
See RINVOQ’s safety data across clinical trials

Explore RINVOQ data in gastroenterology
For moderate to severe Crohn's disease (CD) or moderate to severe ulcerative colitis (UC) in adult TNFi-IR patients1